Study Overview
Let's Talk About It




The Time is Now
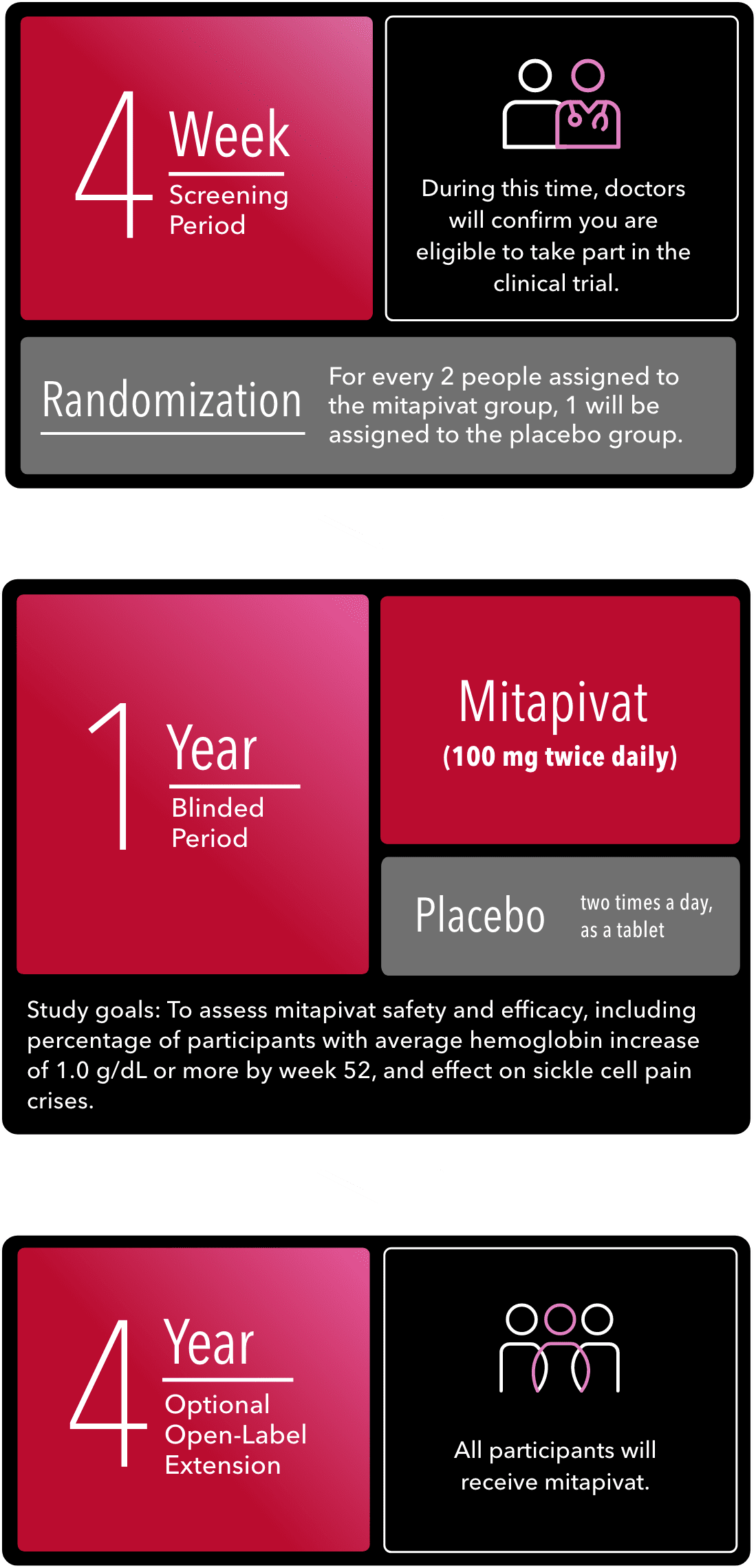
Mitapivat is a potential new therapy for the pain crises and anemia of sickle cell disease. There have been two studies for mitapivat so far — a smaller phase 1 study and a phase 2 study. Now the next part of RISE UP, a phase 3 study, is enrolling Warriors.
PHASE 3 (Now Enrolling)